|
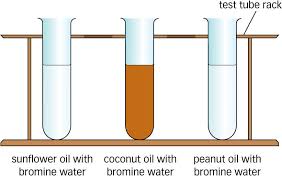
In organic chemistry, the bromine test is a qualitative test for the presence of unsaturation (carbon to carbon double or triple bonds) and phenols.
An unknown sample is treated with a small amount of elemental bromine — either as an aqueous solution, or as a solution in dichloromethane or carbon tetrachloride. Unsaturation and/or phenol is shown by the disappearance of the deep brown coloration of bromine when the bromine has been used up by reaction with the unknown sample. The formation of a white precipitate (a brominated phenol) indicates that the unknown was a phenol. The more unsaturated an unknown is, the more bromine it can decolourise.It has more affinity towards carbon with less alpha hydrogens. USEFUL LINKS -- 1)https://www.youtube.com/watch?v=ZXcS3oY9wQo
0 Comments
Leave a Reply. |
AuthorHII FRIENDS I AM NIHAR MORE FOR DOUBTS ,MORE INFORMATIVE UPLOADS. PLEASE CLICK ON READ MORE FOR FULL INFORMATION
|